This web page was produced as an assignment for Genetics 564, an undergraduate course at UW-Madison
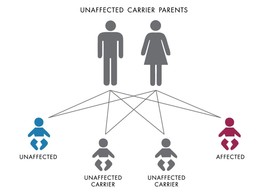 Figure 1. An example of autosomal recessive inheritance.
Figure 1. An example of autosomal recessive inheritance.
Nijmegen Breakage Syndrome, NBS, is a rare genetic condition of chromosomal instability. Because of indistinguishable chromosomal similarities between these patients and patients diagnosed with ataxia-telangiectasia (A-T), this disease is designated as Ataxia-telangiectasia variant-1 (AT-V1). However, NBS differs greatly in the phenotypic aspects of its patients and is characterized by the following [1][2][3]:
Unusually Small Head Size [Progressive Microcephaly]. Typically apparent from birth, growth of the head is out of sync with the body. An appearance in which the head seems to shrink as the body grows is common. Intellectual disability. Although developmental milestones are reached at a normal pace during the first year or two of life, progress begins to decline thereafter. Hyperactivity is sometimes noted during early childhood. Distinctive facial features.Features become more obvious with age and typically include: sloping forehead, large ears, small jaw, prominent nose, and up-slanting palpebral fissures (upward pointing outer eye corners). Short stature. Growth begins at a slowed rate throughout infancy and childhood, eventually returning to a normal rate. Potential height is therefore never reached. Recurrent respiratory tract infections. Immunodeficiency, or a "broken" immune system, is common. This results in T cells, B cells and specific proteins of the immune system (immunoglobulin G and A) to reside at abnormally low levels. Increased risk of cancer. Individuals with this disease are 50 times more likely to develop cancer; non-Hodgkin lymphoma being the most common. Additionally, brain tumors and cancer of the muscle tissue are also seen. Premature Ovarian Failure in Females. Premature ovarian failure results in infrequent menstruation or primary amenorrhea (not beginning menstruation by age 16). Typically, women with this disease are infertile and unable to reproduce. A variety of additional symptoms are commonly seen in NBS patients [2]. This disease, known to have an increased incidence among populations of Eastern Europe and Slavic, affects an estimated 1 in 100,000 infants [1][2]. Inheritance of NBS depends on the genetic makeup of the individual’s parents. NBN, the only gene currently connected with this disease, is altered in nearly 100 percent of those who are affected. Because the disease is autosomal recessive, each parent needs to posses a mutation of the gene and subsequently pass it on to the child [3]. There is a 25 percent chance a child will develop NBS if both parents are carriers for the disease (Figure 1). These carriers, though no apparent symptoms have been reported, are thought to have a higher susceptibility for various cancers [3]. Diagnosis of NBS relies on the DNA analysis of the NBN gene. If needed, testing for the presence of the nibrin protein by immunoblotting, as well as determining radiosensitivity using a colony survival assay can further verify the diagnosis [3]. Clinical Diagnostic Flow-chart for NBS. Prognosis of NBS is still quite poor. Around 50 percent of patients will develop non-Hodgkin lymphoma before the age of 15 [1]. Cancer development is responsible for the majority of fatalities, though death also commonly results from infections that ultimatly lead to renal or liver insufficiency, bone marrow aplasia, or respiratory failure [4]. The life expectancy for those affected by NBS varies greatly, but is not normally beyond 30 or 40 years [5]. Treatment surrounding NBS is not concrete. At this time, there are no direct therapeutic regimens for the disease. However, there exist a variety of treatments to help reduce its symptoms [4]. The mitogenic features shared with A-T patients, including spontaneous chromosomal instability, clonal occurrence of rearrangements of chromosomes, chromosomal and cellular hypersensitivity to irradiation, and radioresistant DNA synthesis, limit the patients ability to utilize chemotherapy and eliminate their option of radiation therapy [2]. Research is continuously developing, and in the case of those affected by NBS, we may look to "sister" diseases for possible solutions (Video 1). |
|
|
Video 1. Causes and Drug Development in Ataxia Telangiectasia. Dr Richard Gatti.
Retrieved from: Youtube Credit to: ecancer
Retrieved from: Youtube Credit to: ecancer
|
NBN (nibrin) is a 4,639 base pair gene located on chromosome 8, specifically, 8q21.3 [Accession: NM_002485.4, FASTA]. The cell uses information from this gene to code for a 754 amino acid, 95kDa protein [Accession: NP_002476.2, FASTA]. This protein is commonly referred to as p95 or NBS1.
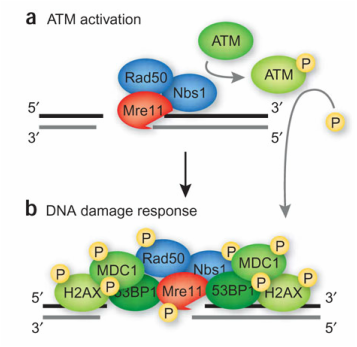
A mutated NBN gene is currently the only known commonality in patients with NBS. Under normal conditions, the protein is involved in crucial cellular functions. One such function is assisting in the repair of damaged DNA. Working with two other proteins, MRE11A and RAD50, NBN contributes to a complex called MRN. Specifically, nibrin is responsible for transporting the MRE11A and RAD50 inside the nucleus and to the site of DNA damage where mending of the break will then begin [1]. Once in position, the complex activates the ATM kinase, responsible for coordinating repair of the DNA [6](Figure 2). Because of this NBN can be considered a tumor suppressor protein for, without this process, cells can accumulate errors that may lead to cell death or uncontrolled growth [1]. Specifically, in those with NBS, mutations of the NBN gene lead to a truncated or abnormally short version of the protein. Today, ten, if not more, mutations of NBN are known to lead to NBS. Of all |
|
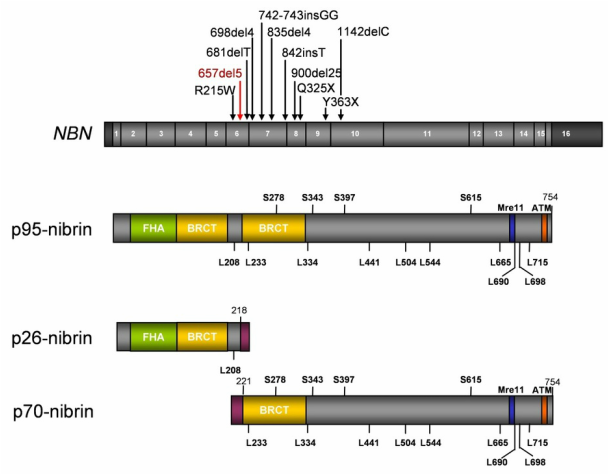
documented cases, a mutation involving a deletion of 5 nucleotides in the 6th exon of the gene (c.657_661del5) has proven most common. Because of this deletion, the resulting protein is fragmented into two notably shorter pieces now termed p26-nibrin and p70-nibrin [7](Figure 3). P70, the portion of protein containing sites responsible for interaction with the MRN complex, is essential to maintain life. Though not completely ineffective, the p70 fragment has lowered expression and is not nearly as successful at responding to damaged DNA [1].
|
|
The inability to respond and repair DNA damage is extremely significant to the future of the cell. When genetic damage is not fixed it can build up and trigger the cell to grow and divide irregularly. This increases the risk for those with NBS to develop cancer. In contrast, it is thought that the lack of DNA repair decreases the ability for immune cells to grow and divide, resulting in low numbers of cells and ultimately, a malfunctioning immune system [1].
|
|
References:
Banner Image [Yana]. http://joyrx.org/joyrx/friendship-rx/community-hero-exhibit/2007-hero-kids/yana-brichka.html Figure 1. http://www.testmygenes.info/terms/autosomal-recessive-inheritance/ Figure 2. Zha, S., Boboila, C., Alt, F. (2009). Mre11: roles in DNA repair beyond homologous recombination. Retrieved January 30, 2015, from http://www.nature.com/nsmb/journal/v16/n8/full/nsmb0809-798.html Figure 3. Chrzanowska, K., Gregorek, H., Dembowska-Bagińska, B., Kalina, M., Digweed, M. (2012, February 28). Nijmegen Breakage Syndrome (NBS). Orphanet Journal of Rare Diseases 2012, 7: 13. doi:10.1186/1750-1172-7-13. Video 1. Retrieved from Youtube: https://www.youtube.com/watch?v=eYEQ4eB2j_I Video 1 Credit. ecancer. Interview of Gatti, R. (2011, December 19) Causes and drug development in ataxia telangiectasia. ASH 2011. http://ecancer.org/conference/139-ash-2011/video/1279/causes-and-drug-development-in-ataxia-telangiectasia.php [1] Nijmegen Breakage Syndrome. (2011, April 1). Retrieved January 30, 2015, from http://ghr.nlm.nih.gov/condition/nijmegen-breakage-syndrome [2] McKusick, V., Kniffin, C. (2009, November 2). OMIM Entry- #251260- Nijmegen Breakage Syndrome. Retreved January 31, 2015, from http://www.omim.org/entry/251260 [3] Varon, R., Demuth, I., Digweed, M. (2014, May 18). Nijmegen Breakage Syndrome. Retrieved February 2, 2015, from http://www.ncbi.nlm.nih.gov/books/NBK1176/ [4] Chrzanowska, K., Janniger, C. (2014, Septemper 29). Nijmegen Breakage Syndrome. Retrieved January 30, 2105, from http://emedicine.medscape.com/article/1116869-overview [5] Nijmegen Breakage Syndrome. (2015). Retrieved January 30, 2015, from https://www.counsyl.com/services/family-prep-screen/diseases/nijmegen-breakage-syndrome/ [6] Zha, S., Boboila, C., Alt, F. (2009). Mre11: roles in DNA repair beyond homologous recombination. Retrieved January 30, 2015, from http://www.nature.com/nsmb/journal/v16/n8/full/nsmb0809-798.html [7] Chrzanowska, K., Gregorek, H., Dembowska-Bagińska, B., Kalina, M., Digweed, M. (2012, February 28). Nijmegen Breakage Syndrome (NBS). Orphanet Journal of Rare Diseases 2012, 7: 13. doi:10.1186/1750-1172-7-13. |
|
|